The FDA’s new approach to CSV is Computer Software Assurance (CSA). It represents a step-change in computer system validation, placing critical thinking at the centre of the CSV process. CSA is a risk- based approach to validate a system directly impacting Product Quality and Patient Safety. The traditional approach to Computer System Validation (CSV) has […]
Pharmaceutical manufacturing is one of the most closed regulated industry. The Pharmaceutical industry have last 50 years of admirable quality experience. Vetting of Quality Assurance is perspective meets its regulatory specifications. Compliance can’t take granted if their products are not meeting to their appreciate safety, Quality, requirements, customers satisfactions. Quality control is the part of […]
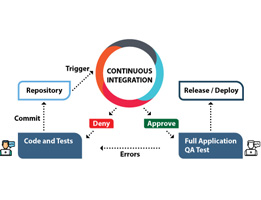
Continuous integration (CI) is a method of integrating changes needed in one single project which is developed, then tested accordingly, this method helps developers to deliver a software in a more efficient and a predictable manner. To understand the importance of continuous integration first we understand how the process is going before continuous integration is […]