The FDA’s new approach to CSV is Computer Software Assurance (CSA). It represents a step-change in computer system validation, placing critical thinking at the centre of the CSV process. CSA is a risk- based approach to validate a system directly impacting Product Quality and Patient Safety. The traditional approach to Computer System Validation (CSV) has […]
Pharmaceutical manufacturing is one of the most closed regulated industry. The Pharmaceutical industry have last 50 years of admirable quality experience. Vetting of Quality Assurance is perspective meets its regulatory specifications. Compliance can’t take granted if their products are not meeting to their appreciate safety, Quality, requirements, customers satisfactions. Quality control is the part of […]
An Electronic Lab Notebook is an Electronic version of the traditional lab notebook. An ELN is a software for documenting your research work and where you can record and store experimental information and results. ELN allow you to find your research data in seconds. It provides functions like your traditional paper notebook, but it does […]
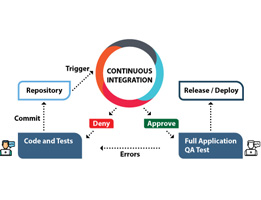
Continuous integration (CI) is a method of integrating changes needed in one single project which is developed, then tested accordingly, this method helps developers to deliver a software in a more efficient and a predictable manner. To understand the importance of continuous integration first we understand how the process is going before continuous integration is […]
What is an enterprise application? The software solution used across departments of an enterprise to improve overall efficiency, effectiveness and productivity of various functions. Such system normally works on single, undisputed source of master data; and modules exhibit usage in multiple departments with interdependency over one-another. What is a successful enterprise application deployment? Those who […]
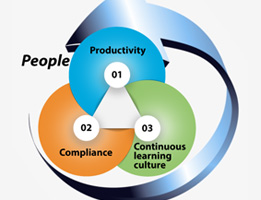
Continuing the discussion about digital transformation strategies in clinical research, the People angle, which would be the most critical angle. Like any other business process reengineering, it is people who will make it success or failure. It is more important in digital transformation, because in effort to make people more powerful and effective, and, empower […]
Each regulated record (is a GMP record) and should be periodically reviewed Strong SOP and Work instructions repository covering all working areas Templates for all documentation, processes – recommended, mandatory VMP for computer systems – mandatory for all who are regulatory audited and use the same for validation across for computer systems URS – always […]
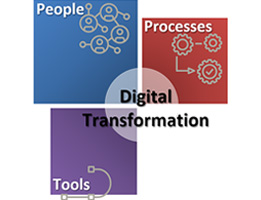
When we talk about ‘Digital Transformation’, there are wide range of definitions – the simplest covers digitization of processes, to more complex one that talks about complete revamping and reimaging the business in the era of digital technologies. Of course, in terms of clinical research; when we talk about digital transformation; we are not thinking […]
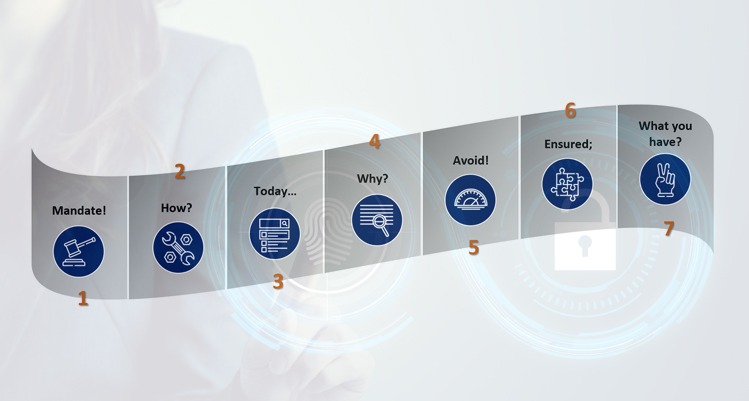
CSV is mandatory for implementation of software systems in a GxP regulated environment. And, it would incur huge costs if not handled adequately. With increasing number of software systems implemented in any organization, it becomes utmost important to check the costs at every step, especially of CSV. The CSV tools include the following: Mandate! To […]
Memory management is a very vast concept and each individual can have different idea or way to implement this in different programming language. In this blog, we are going to see the details of memory management in respect to .NET. Well, .NET is one of the fantastic piece of software engineering. With this framework, user […]